Many people think twice about the idea of having germicidal UV running in the upper area of occupied rooms, concerned about possible harm to human health. However, decades of in-field data and a leading study show that the actual risks are insignificant.
How does UVC light work and what are the risks?
UVC light is found within 100-280 nm range of the ultraviolet (UV) light spectrum, with most germicidal bulbs emitting at 254 nm. At this frequency, UVC irradiation has been proven to break down the DNA and RNA of all known miscroscopic pathogens, meaning they cannot replicate and cause the spread of disease.
Upper-room ultraviolet germicidal irradiation (UVGI), or germicidal ultraviolet (GUV), involves installing wall or ceiling-mounted germicidal UV lights in the upper stratum of a room, where they disinfect exhaled air. Unlike other GUV applications like UV robots and surface sterilization lamps, they are designed to operate while people are present. At high intensity for prolonged periods, exposure to UVC light can cause irritation and swelling to human skin and eyes.
The main risk to people posed by germicidal UVC is prolonged exposure to the eyes, which can result in a condition called photokeratoconjunctivitis – inflammation of the cellular outer layer of the cornea.
The skin irritation is called photodermatitis, which is similar to sunburn. Because UVC light is nearly completely absorbed by compounds called chromophores found in the outer layer of dead skin, this is less of a concern.
Upper-room UV air disinfection: a long history of success with few cases of injury

You might be surprised to learn that upper-room UVC was first used as early as the 1930s, initially in hospitals to prevent cross-infection, and later in schools to combat measles transmission. From the 1950s through to the 1970s much of the upper-room UVGI research and development was focussed on fighting tuberculosis (TB). In the late 1980s public health authorities across the United States – faced with a surge of antibiotic-resistant TB combined with the HIV epidemic among vulnerable populations – began to scale up use of upper-room UVGI in homeless shelters, with use continuing to this day. The landmark study on the safety of upper-room UVC – Safety of Upper-Room Ultraviolet Germicidal Air Disinfection for Room Occupants: Results from the Tuberculosis Ultraviolet Shelter Study (TUSS) – was produced by the National Center for Biotechnology Information (NCBI), based on field work carried out in 14 homeless shelters across 6 US cities from 1997 to 2004.
In providing context, the TUSS report details all known documented cases of accidental human UVC injury prior to the study. It is important to note that of these five instances, only one involved upper-room UVGI that is could be described as “properly installed”, with the other four involving lower room applications and/or varying degrees of human and technical error:
- In the 1970s eight hospital workers developed photokeratoconjunctvitis after 20 minutes of exposure to a high-intensity, lower-room UVC light fixture that was being used above an operating room table. It had been mistakenly left on after a surgical procedure and the staff who became exposed were not wearing appropriate personal protective gear;
- Two nurses and a housekeeper in a Botswana hospital experienced facial peeling and eye discomfort after they inadvertently switched on an unshielded 36W UVGI lamp. It had been left installed in an area which had been converted from a sputum induction room to an office. The on/off switch for the UVC bulb was right next to a switch for a normal fluorescent light. At the UVGI levels emitted by the light, the threshold limit value (TLV) for 254 nm UV would have been met in a matter of minutes. Their symptoms resolved completely in two to four days;
- In 2006 in Italy, 26 medical students observing an autopsy were exposed to a bare UVGI bulb for 90 minutes due to a malfunctioning timer that was meant to ensure the UVC light only activated when the room was unoccupied. Despite meter measurements indicating they would have experienced exposure levels 20 to 100 times the TLV, skin symptoms resolved within two weeks and ocular symptoms within two to four days;
- In the most extreme case documented, significant human error was involved. A UVGI tube was installed in place of a normal fluorescent one in a desk fixture in the ward of a psychiatric hospital. Over a four-month period, 58 people reported significant eye symptoms. Because the room occupants were so close to the UVGI lamp, they would have quickly exceeded the tolerable 254nm dose;
- During the resurgence of TB across in the mid 80s to early 90s, commercially available UVGI luminaires tended to produce excessive levels of UVC irradiation in rooms with lower, UV-reflective ceilings. Staff at some health facilities and shelters reported eye and skin irritation during the work week that improved over the weekends. Photometry tests revealed UVC irradiance levels 10 times higher than was expected, which led to the development of the tightly louvered UVGI fixtures that are commonly found today.
Louvers were added to open UVGI systems first developed in the 1940s, when building typically had ceiling heights in excess of 3m, to better suit modern architecture and comply with widely accepted peak irradiance levels at average eye level (1.68m) of around 0.2 μW/cm2. This came at a cost to efficacy, as these louvers also blocked out sections of the germicidal UV beam and created ‘blackspots’ were airborne pathogens could hide.
The TUSS test: upper-room UVGI worries are 'all in the mind'?
The TUSS data is especially valuable because it is the result of a properly conducted double-blind, placebo-controlled trial. Throughout the 7 years of study, an unblinded head of data safety and monitoring would assign each of the 14 shelters either an active or placebo status. Each shelter had a specially designed upper-room UVGI system installed that could be toggled from ‘active’ to ‘placebo’ state either by replacing the UVGI bulb with a dummy unit, or inserting a piece of glass – impenetrable to UV – in front of the light.
Study participants – both clients and staff of the shelters – were completely unaware of whether they were in an active or placebo phase at any given time. Data was gathered via interviews conducted with safety questions focussed on eye and skin symptoms. Of 3,611 interviews conducted over the 7-year study, there were 223 reports of an eye or skin symptom. In most cases, it was possible to determine whether the symptom occurred during an active or placebo period, although in some responses the period was unclear.
Ultimately, the data revealed that the incidence of reported symptoms occurred at the same rate during the active period as the placebo period. This indicates that any discomfort was psychological, or due to unrelated conditions like eczema or bacterial skin infections, rather than a real physiological reaction to upper-room UVGI.
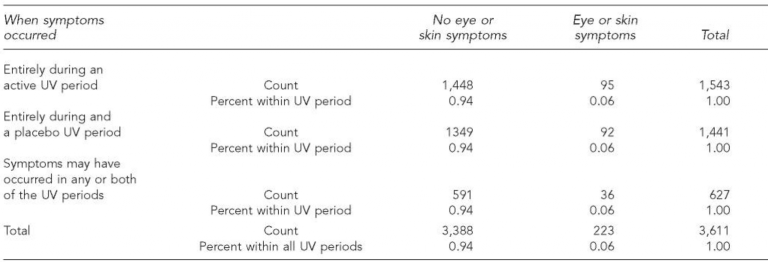
During the study, there was one definite case of photokeratoconjunctivitis, which resulted from human error. A double bunk bed was substituted for a single bed in a shelter after the upper GUV system had already been designed and installed, exposing the upper bunk occupant to higher than tolerable UVC levels. This issue was resolved by moving the UV light ficture higher up on the wall.
Testing the practicality of accepted TLV - more (or less) than meets the eye
As discussed, TLV is a defined value of tolerable UVC irradiation that is based on assumptions of people’s eyes being continuously exposed to the UV light source. The widely circulated standard is for upper-room GUV installations to give off no more than 0.2 μW/cm2 (UVC power) at average eye level (~1.7m), which would allow for 8 hours of daily exposure before the total exposure threshold of 6mJ / cm2 is reached.
The authors of the TUSS study have described the 0.2 μW/cm2 benchmark as “inappropriately low”, pointing to a study conducted into the actual exposures experienced by a range of subjects across multiple upper-room UVGI settings. The study involved placing body-worn UVC light meters on the upper chest of participants and then comparing the actual UVC doses they experienced to the calculated doses (peak eye level UVC for 8 hours) for each setting.
The study revealed that even the more ‘at-risk’ participants – ie those who occupied rooms with higher peak UVC readings at eye-level and who spent much of the day on their feet – recorded actual UVC doses that were a mere fraction of the TLV of 6mJ / cm2, with an average of just 9.1% across the 19 subjects.
Graphs showing the UVC power in μW/cm2 for each subject show that for much of the time, the exposure level is very low, only spiking at particular points of the day, when the subject might look directly at the light source for example.
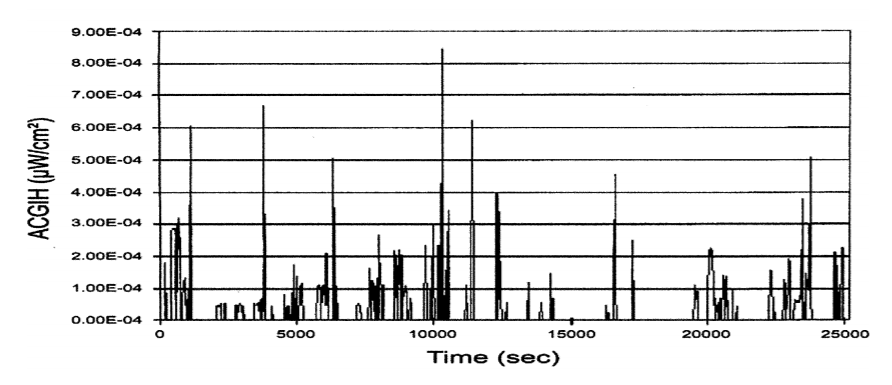
Although it wasn’t practical to place a UV light meter at actual human eye level, the upper chest was deemed an appropriate surrogate for approximating the way the eye is shielded from a light source by the contours of the face and a person’s natural movement within a room.
The findings are significant because they reveal that the widely accepted parameters are arguably too cautious, and that by adhering to them upper-room UVGI installations are not as germicidally effective as they might otherwise be.
Upper-room UVGI has been deployed in various forms for the past 80 years and there have only been a handful of cases of human injury attributable to the technology. When properly designed, installed and tested, upper-room germicidal UV systems are proven to be safe and effective.
In order to guarantee safety, modifications have been made to adapt germicidal UV to the challenges posed by evolving building characteristics like lower ceiling heights and UV reflective surfaces. If anything, our collective approach has been overly cautious. Ironically, an effort to mitigate against the risks of UVC-related eye or skin irritation might result in the increased risk of illness due to germicidal inefficiency.
